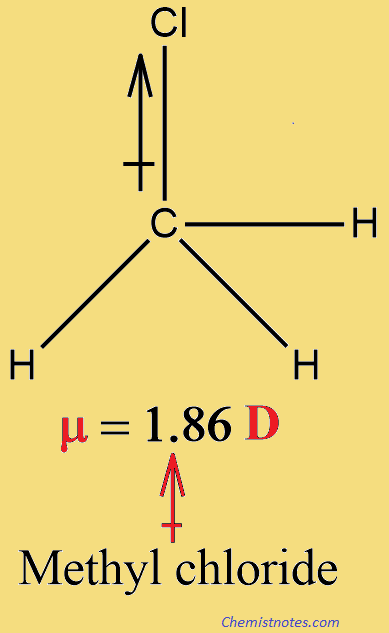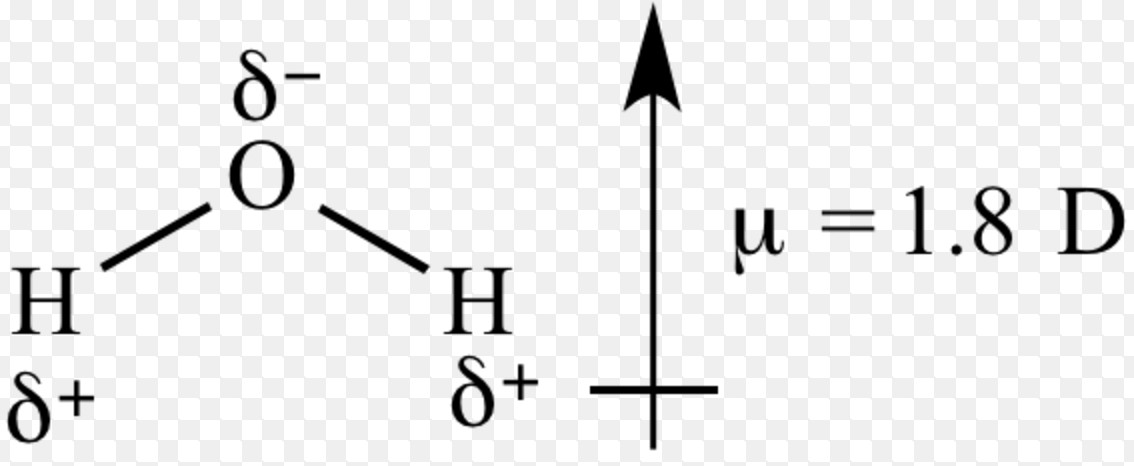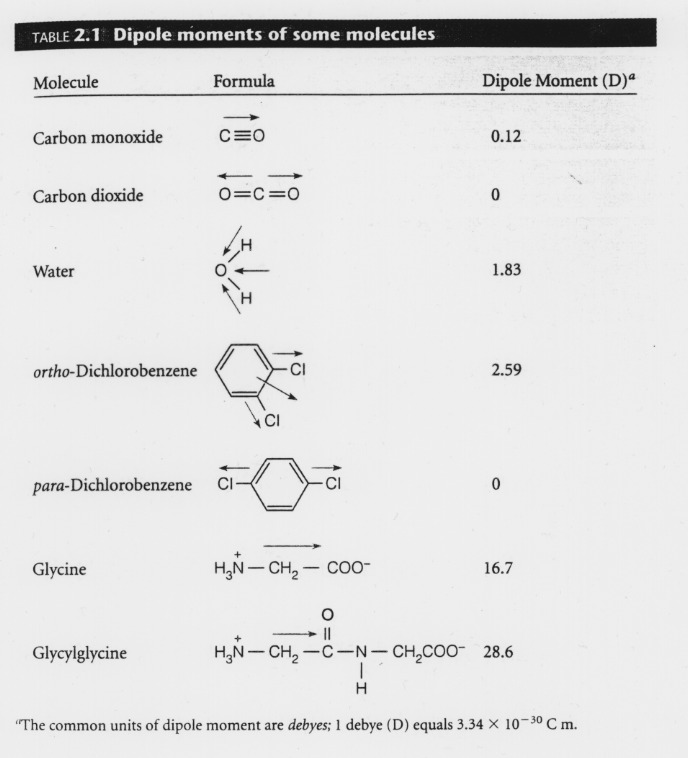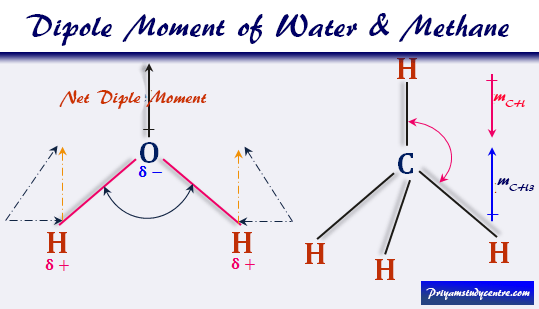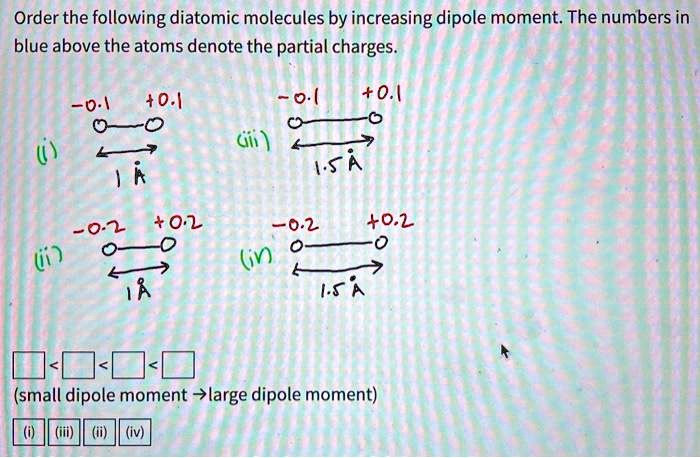
SOLVED: Order the following diatomic molecules by increasing dipole moment: The numbers in blue above the atoms denote the partial charges 0: +0.| 0 0 U) +0. Gi; ) 0-2 +0.2 +0.2 @) (v) LSA (small dipole moment large dipole moment) 0. (
The orientation of the molecule and of its electric dipole moment in... | Download Scientific Diagram









/chapter3/pages3and4/page3and4_files/cnoorbital.png)