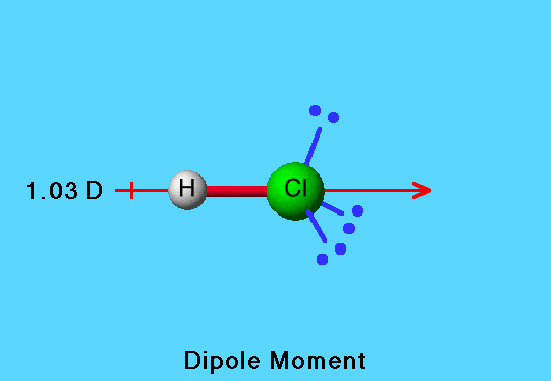
18. Dipole moment ofHCL=1.03D,HI=0.38DHCL=1.03D,HI=0.38D. Bond length ofHCL=1.3AHCL=1.3AandHI=1.6AHI=1.6A. The ratio of fraction of an electric charge,δ δ , existing on each atom in HCl and HI is : (1)1.2:1(3)3.3:1(2)2.7:1(4)1:3.3
Why is it so that the dipole moment of HI < HBr< HCl<HF, where HI is lowest and HF is highest? - Quora

An HCl molecule has a dipole moment of `3.4xx 10(-30) Cm` . Assuming that equal and opposite cha... - YouTube
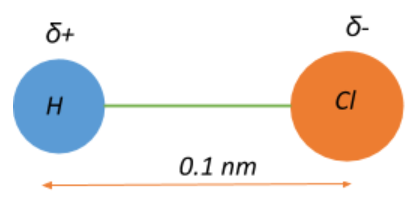
The electric dipole moment of an HCl atom is $3.4 \\times 10^{-30}\\;Cm$. The charges on both atoms are unlike and of the same magnitude. Magnitude of this charge is ______. The distance

TAMLIQ BJ VILIVM LIQUI MV LIQ The electric dipole moment of an HCL atom is 3.4 x 10-30 cm. The charges on both atoms are unlike and of same magnitude. Magnitude of

The observed dipole moment of `HCl` is `1.03D`. If the bond length of HCL is `1.3Å`, then the - YouTube

SOLVED: Dipole moment (μ) Polarizability volume " HCl 1.08 D 2.63x10^-30 m^3 HI 0.42 D 5.45x10^-30 m^3 Q1. A) For a given electric field, what is the ratio of the induced dipole

SOLVED: Estimate the dipole moment of an HCl molecule from the electronegativities of the elements and express the answer in debye and coulomb meters (C m). Answer: 0.9 D = 3.00×10^-30 C

Dipole moment of HCl molecule is found to be 0.816 D . Assuming HCl bond length to be equal to 1 A , the
the observed dipole moment of hcl is 1.03D bond length is 1.275A then the precentage of ionic character is



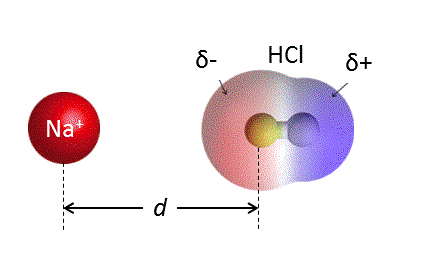
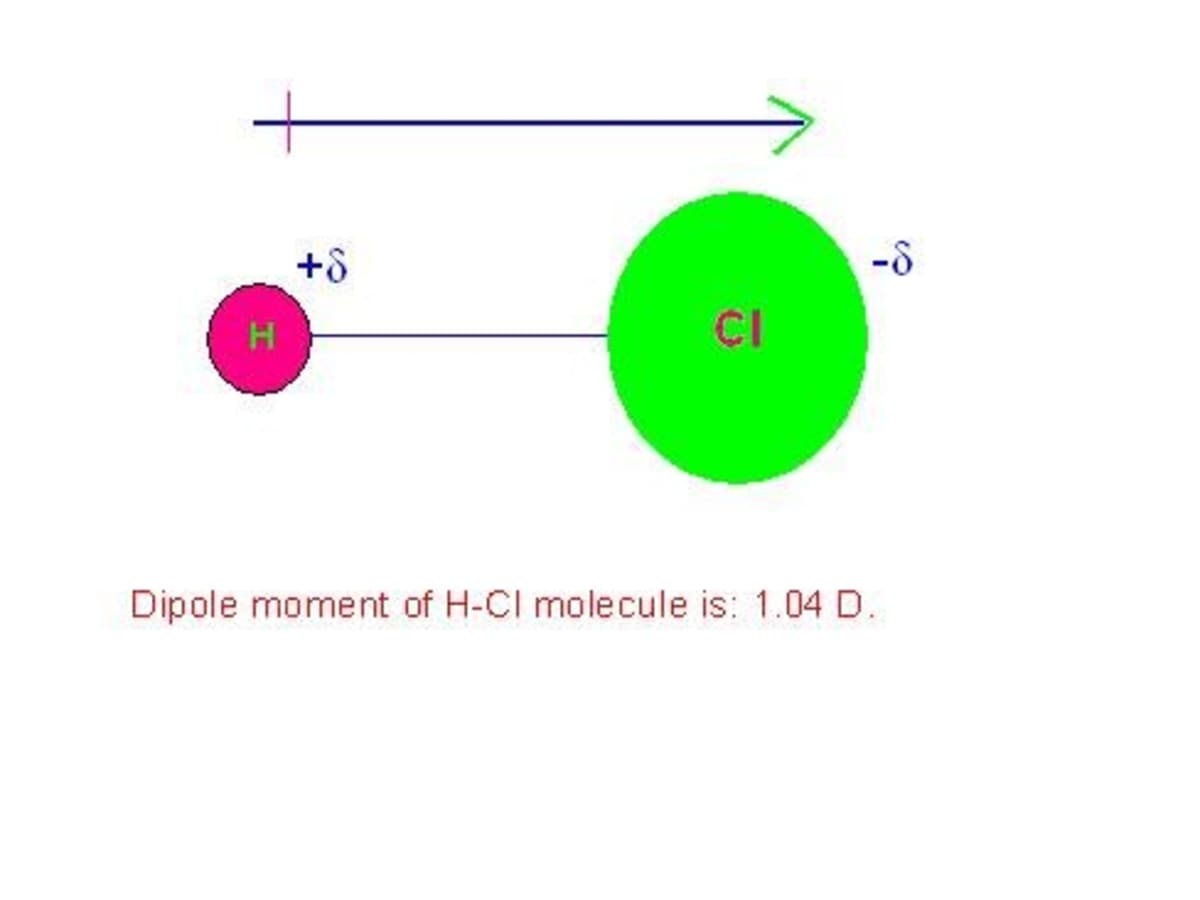

![[Bengali] Calculate the % ionic character in HCI molecule. Given bond [Bengali] Calculate the % ionic character in HCI molecule. Given bond](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/8903493.webp)


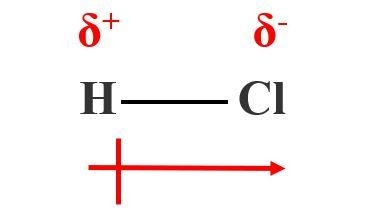
![Best Overview: Is HCl polar or nonpolar [#1] - Science Education and Tutorials Best Overview: Is HCl polar or nonpolar [#1] - Science Education and Tutorials](http://sciedutut.com/wp-content/uploads/2021/06/HCl-cover-banner.png)